Tag: pharmacokinetics
Modified-Release Formulations: What You Need to Know About Bioequivalence Rules
Posted by Ian SInclair On 18 Dec, 2025 Comments (0)
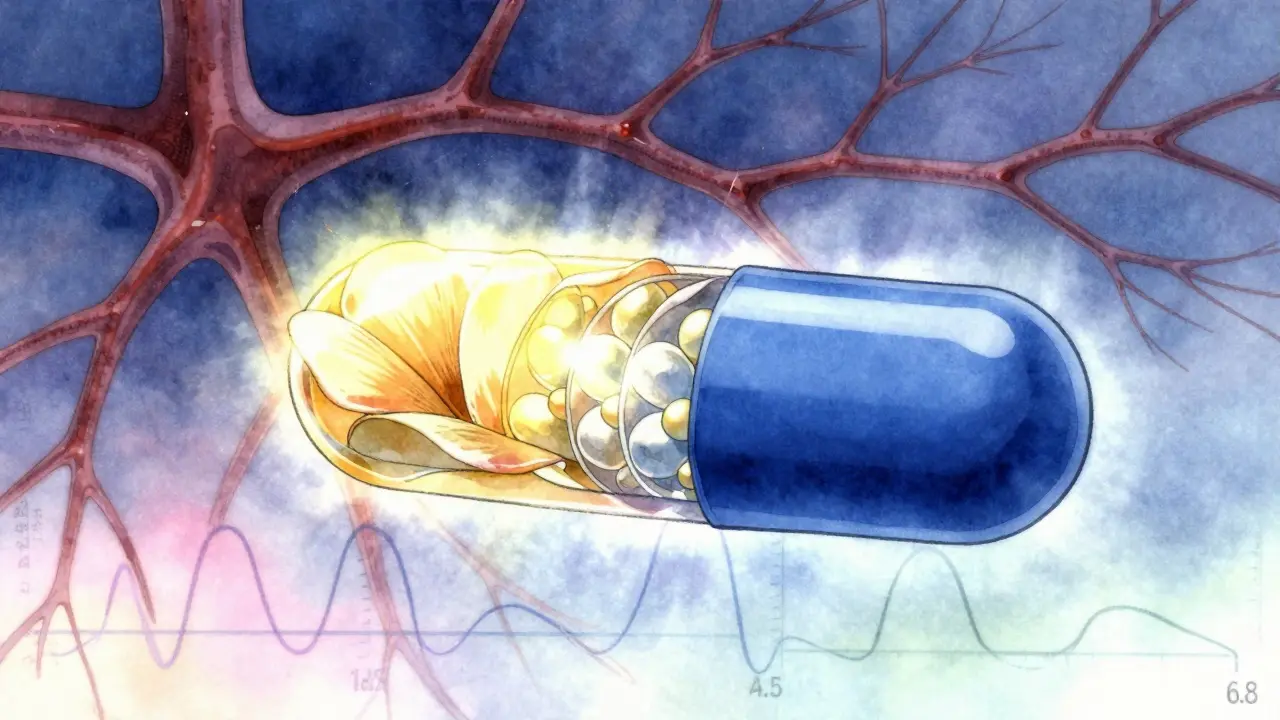
Modified-release formulations require special bioequivalence testing because timing and rate of drug release matter. Learn how regulators like the FDA and EMA ensure generics work the same as brand-name drugs.

Losartan Hypersensitivity: Causes, Genetics, and Key Risk Factors
May 19, 2025 at 04:19

How to Get Enough Medication Before International Travel: A Practical Guide
November 21, 2025 at 11:06

Does Fluoxetine Aid in Weight Loss? Exploring its Usage Beyond Depression
March 22, 2024 at 01:50

Amebiasis and Its Impact on the Global Economy: The True Cost of Infection
May 14, 2024 at 15:46

Baby Planning: How to Prepare Your Home for a Newborn
May 13, 2023 at 00:08
