Tag: ER tablets
Modified-Release Formulations: What You Need to Know About Bioequivalence Rules
Posted by Ian SInclair On 18 Dec, 2025 Comments (0)
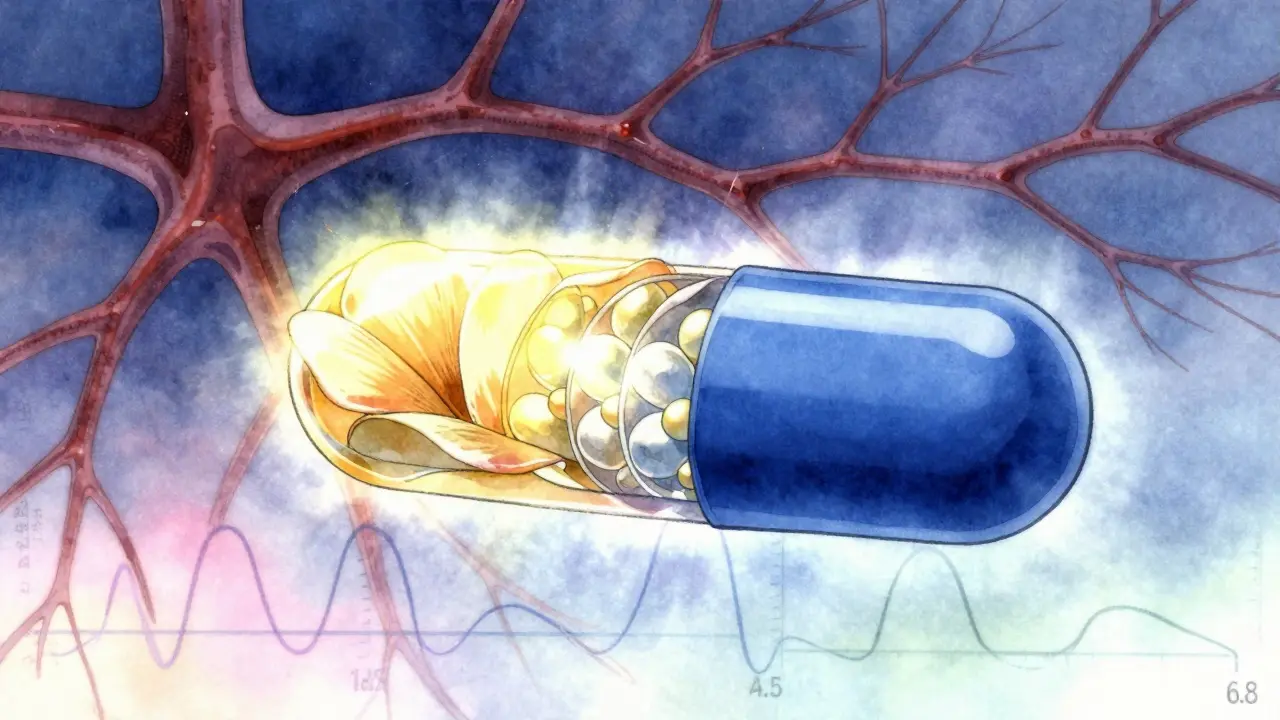
Modified-release formulations require special bioequivalence testing because timing and rate of drug release matter. Learn how regulators like the FDA and EMA ensure generics work the same as brand-name drugs.

How to Buy Cheap Generic Yasmin Online Safely
September 28, 2025 at 20:58

How to Audit Your Medication Bag Before Leaving the Pharmacy: A Simple 7-Step Safety Check
January 25, 2026 at 23:09

Effexor XR vs. Top Antidepressant Alternatives: A Detailed Comparison
October 8, 2025 at 13:43

Corticosteroids and Diabetes: How Steroids Cause High Blood Sugar and How to Manage It
February 1, 2026 at 11:57

Dilantin (Phenytoin) vs Alternative Antiepileptic Drugs: Detailed Comparison
October 17, 2025 at 21:48
