Tag: modified-release formulations
Modified-Release Formulations: What You Need to Know About Bioequivalence Rules
Posted by Ian SInclair On 18 Dec, 2025 Comments (0)
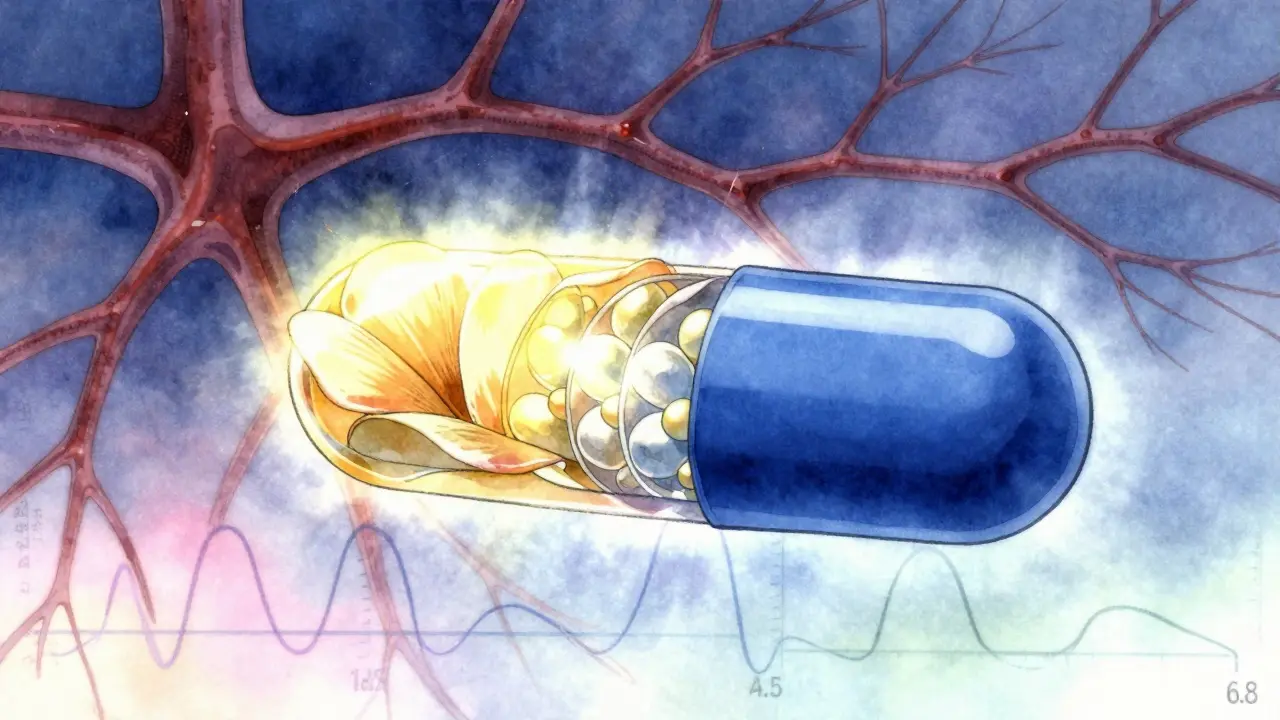
Modified-release formulations require special bioequivalence testing because timing and rate of drug release matter. Learn how regulators like the FDA and EMA ensure generics work the same as brand-name drugs.

Thyroid Eye Disease: Symptoms, Steroids, and Biologics
March 26, 2026 at 12:18

Emotional Blunting from SSRIs: What It Is and How to Fix It
November 24, 2025 at 15:22

How to Buy Cheap Generic Metformin Online Safely
October 13, 2025 at 19:04
The Role of Assistive Technology in Empowering People with Intellectual Disabilities
July 16, 2023 at 04:32
Avoiding Hemolysis: G6PD Deficiency and Medication Safety Guide
April 4, 2026 at 00:24
